- Blog
- Connect webook to get full list dialogflow
- Pro tools 2018 torrent mac
- Argen y tina
- Inkdrop snowflake
- Gw2 ad infinitum guide
- Nightlife puerto viejo costa rica
- Which atom has the smallest atomic radius
- Negative symptoms of schizophrenia
- Sunken treasure chests archeage map
- Keyboard shortcut to make my screensaver start
- Adobe lightroom classic cc unable to use cityblue font
- Ishowu audio capture not working
- Mp3 joiner mp3 splitter

Energy gets the atomic size of all the elements in this period. And the authority says this is greater than that. You sit down with atomic size increases so mean. 119 went first will come somewhere here and want you to be here.
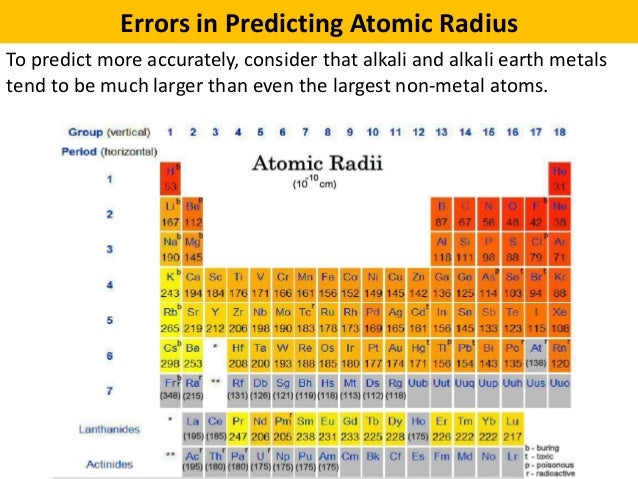
When you look at the periodic table a human this is what you have here. Because Elements 1 20 Is beneath elements 1 16. But elements 116 is not larger than elements 111 23. Therefore elements 119 is larger than element 1 20. So any discovery was send it in this group 11 night then after 11 night to the left of one to the right of 119 will be elements 1 20 two. From uh basic knowledge of the variation of atomic size across the period. Which I know you have been discovered and elements 120. We were given element 116 Which is live memorial elements 119. The smallest radio ceo clothing Easy enough right now we want to get 3rd one to order see With AC. Really done a pretty table with a lot of short added onto it is very, very big.
#Which atom has the smallest atomic radius series#
Anyway, time to to be way given chlorine, Even chlorine and lantana with Atlanta night, who's in a series of land tonight. So the smallest of the first sets, which is one is helio. And when you're moving you meet hydrogen first before you meet helio making helium smaller than hydrogen. How do you do this higher? Sorry? Everything is larger than helium because from left to right, atomic size decreases. And helios us to lecturers and they are in the same period. So hydrogen and helium hydrogen has to, sorry, hydrogen has only one electron Or is actually a brutal yeah, early one. Okay, so let's look at the first two that were given. So generally atomic sized increases down the group and decreases across the period. Leading to a decrease in atomic size and now when you come to the group down the group more shows are added or electrons move into differentials down the group leading to an increase in atomic sites. So are the electrons move into the same show? There is a correspondent increasing nuclear charge when this increase okay causes the atoms to shrink. You have the same show shows are not added. That's when you move across secured shows are not added. I mean from left right attack sized decreases and the reason is that from left to right there is no addition of cells. This results in and decrease in atomic size. On a purely table from left to right electrons move into the same show with the corresponding increase in nuclear charge. Now let's take a liberation of atomic size and the reality. So with these two ideas in mind you can comfortably choose between a given set of items. Smaller that is the number of protons also known as the nuclear charge and also the number of cautious. In a global in scale atomic size of radio depends on two basic stuff in order to determine the actual it's bigger. Atomic size of atomic radios is defined as how the internet clearer distance between two identical atoms. First off you need to understand the definition of atomic size of radios. So in this question we've been given a list of autonomous elements and irons and have been asked to choose from the group was the one with the smallest radios.
- Blog
- Connect webook to get full list dialogflow
- Pro tools 2018 torrent mac
- Argen y tina
- Inkdrop snowflake
- Gw2 ad infinitum guide
- Nightlife puerto viejo costa rica
- Which atom has the smallest atomic radius
- Negative symptoms of schizophrenia
- Sunken treasure chests archeage map
- Keyboard shortcut to make my screensaver start
- Adobe lightroom classic cc unable to use cityblue font
- Ishowu audio capture not working
- Mp3 joiner mp3 splitter
